SPC software: Smart detection of product and process defects
Is your production working at optimal cost? How to detect possible deviations as they arise
Statistical Process Control (SPC) is a classic. For decades, the power of measurement technology has been growing – and with it the amount of test data. The complexity of production lines is also increasing, as is the number of variants.
Irrespective of this, the demand to proactively clarify quality deficits is increasing. If you want to do justice to all this, you should automate statistical process control with a dedicated SPC software: from data acquisition and measured value analysis to the resulting action management. Our predictive SPC intelligence software solution offers suitable algorithms and user interfaces for each of these tasks.
Advantages of our SPC software
Get to know our use cases

Would you like to find out more about SPC software based on a user example?
Then read through our “Customer Use Case: Fiberline” and get to know the functions of the software even better.


Functions of our SPC software
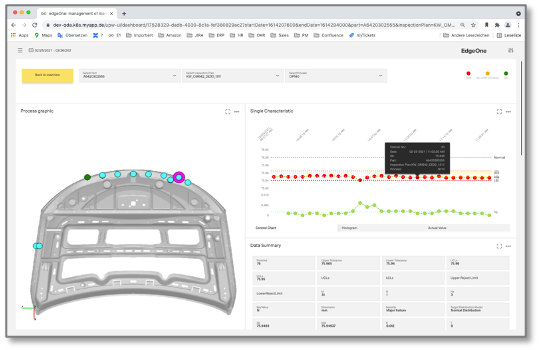
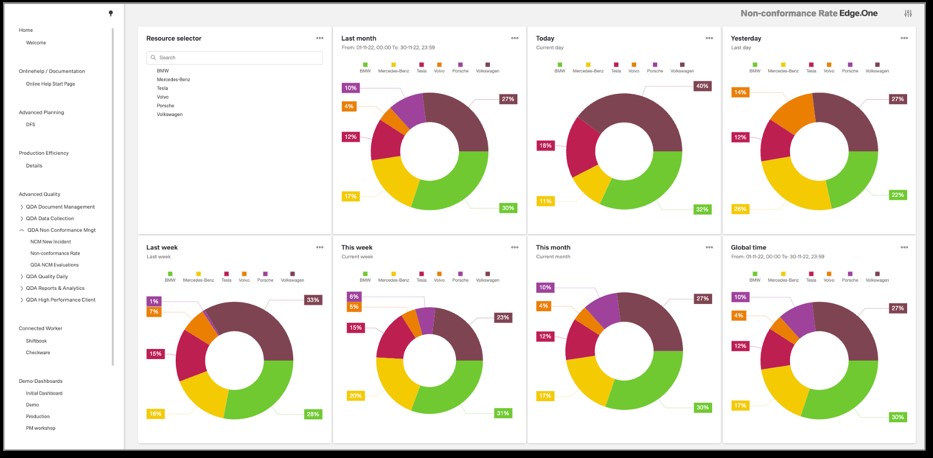
Real-time data from all measuring stations on a single analysis platform
Our SPC software has an architecture in which the inspection technology of your production lines can be completely integrated. The readout of QM data is fully automated and in real time. In this way, our data acquisition software makes the functionality of each individual production line visible, analyzable and optimizable. For this purpose, the measurement data is enriched with suitable metadata, such as identification numbers and tolerance limits. If you also want to use our machine learning algorithms, you can assign any number of context data (e.g. temperature and pressure curves) to the measurement data. In this way, new options are continuously available to uncover error patterns and proactively eliminate their causes.
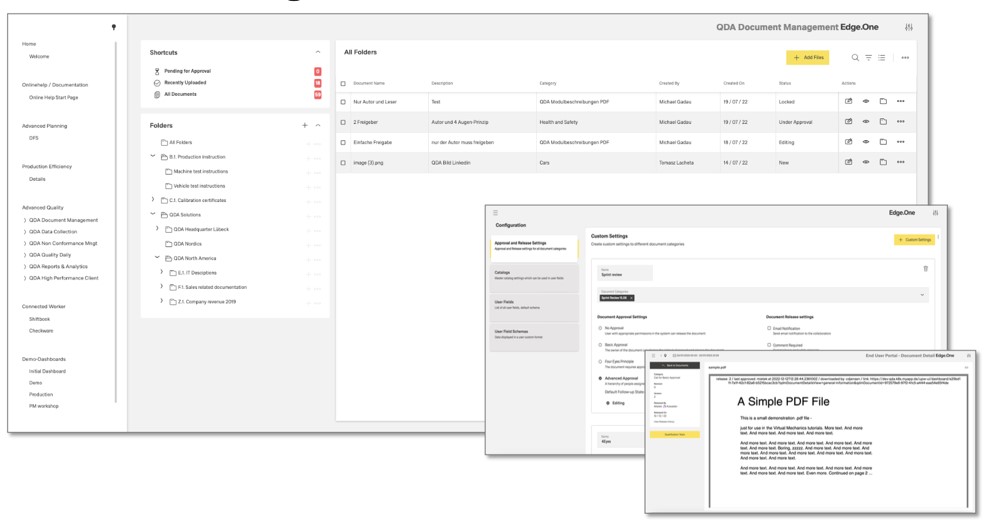
Create QM analyses with the No-Code Report Designer
Democratize reporting and metrics creation! With Report Designer, end users can make their own analyses. They get a drag-and-drop editor to design reports in the way that best suits their information needs. Whether as a control chart, capability study, probability network, histogram, regression or correlation. In addition, the Report Designer makes suggestions for further analyses, e.g. flop 10 overviews for the daily shift discussion. Test certificates are also created fully automatically and distributed to the relevant addressees in the value chain.
Intelligent workflows ensure value creation
The control charts of our statistical process control show the current analysis progress and serve as a jumping-off point to control quality in the production lines. Fully automatically, the statistical process control notifies the responsible value-added participants when test values exceed or threaten to exceed the intervention limits. And should the quality assurance measures actually come too late, the process control also supports the XRM workflows that are now being implemented. This is the case, for example, in claims management, where NCM forms are pre-filled with the relevant measured values. Compared to purely manual claims processing, this reduces the effort required by up to 75%.
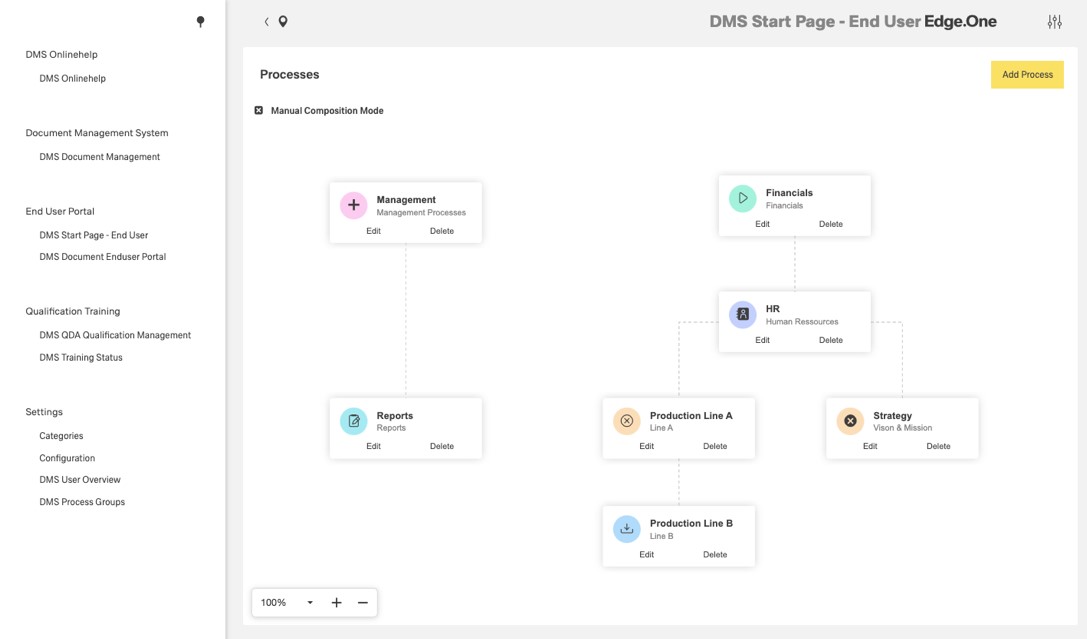
Test plans use existing engineering knowledge
Our inspection planning selects the quality characteristics to be measured on the basis of rules and uses them to create measurement instructions that are practically oriented to the course of the manufacturing process. Manual input of nominal values, tolerances or drawing data by the inspection planner is thus no longer necessary. Instead, our algorithms evaluate the existing CAD, PDM, ERP, PPS and MES data fully automatically. In addition, the SPC software synchronizes with the measurement and gauge management, which is also part of our CAQ suite QDA. This way, you can always ensure that your measurements are performed with correctly calibrated test equipment.
Integration of SPC software
Predictive SPC Intelligence synchronizes with your IT along the entire value chain
Our statistical process control can be used both on premise on your own servers and as a SasS solution in the private, hybrid or public cloud. For user companies that want maximum scalability, availability and data security, we recommend using technology-leading cloud hyperscalers such as Amazon, Azure, Google or the German provider Ionos. For additional security, we provide regular backups that our statistical process control creates on-site at your company.
Thanks to its Kubernetes architecture, the data collection software works platform-independently. In the scope of delivery, you will find a variety of intelligent algorithms with which you can connect your measurement equipment in a fully automated manner. The same applies to the integration of your business software, especially in the areas of CAD, PDM and MES as well as ERP, CRM and SCM.
Since our Statistical Process Control can be used as a SaaS solution, you can reduce the infrastructure and personnel costs of your IT organization to a minimum. The more sites that access the application, the greater the savings. And: Predictive SPC Intelligence supports the standard-compliant assignment of access rights. Conveniently, as soon as users, roles and authorizations have already been created in another IT system, the automatic transfer of this information is also possible.
FAQ: SPC software
What is statistical process control (SPC)?
Statistical Process Control (SPC) is a mature process used by industrial companies to ensure that the output of current manufacturing meets product development specifications. With this in mind, statistical process control provides metrics and reports that can be used to verify the quality of both products and value-added processes in real time.
What tasks does SPC software perform?
The spectrum of applications ranges from the detection of factual quality deficiencies to the prediction of deficiencies that are just beginning to emerge. The latter applies to both production (predictive quality) and maintenance (predictive maintenance). In addition, SPC knowledge serves as an important source of information for driving the Continuous Improvement Process (CIP). The aim of CIP is to identify starting points for companies to sustainably increase the performance of their value creation organization. A well-oiled SPC machine is able to continuously contribute new ideas.
What should SPC solutions be able to do?
Key requirements for statistical process control are:
- automatically connect and evaluate the measurement and control technology of the user company
- bidirectional data exchange with business IT (especially CAD, PDM, PPS and MES as well as ERP, CRM and SCM)
- Provision of analysis tools that can be used intuitively by end users without programming knowledge
- Fully automated creation and distribution of VDI-compliant test plans
- Providing jumping-off points to start QM workflows directly from data analysis and support them end-to-end.
Who benefits from using a fully integrated SPC solution?
Numerous factors contribute to keeping quality costs to a minimum. Among the most important aspects are
- less scrap including the associated disposal costs
- less rework
- fewer returns or recalls
- fewer complaints
- less or no damage to reputation.
At the top of the credit side of SPC are therefore:
- higher profitability of the production goods
- higher productivity of employees
- higher employee satisfaction
- better compliance with the brand promise
- thus higher customer satisfaction and stronger customer loyalty
- promotion of the continuous improvement process.
What’s the difference between Advanced Planning & Scheduling and production scheduling?
Production planning is divided into different planning horizons – strategic, tactical and operational. Depending on the industry and the production process, the time horizons may vary considerably. Advanced Planning & Scheduling (APS) often refers to the strategic-tactical planning, whereas production scheduling describes the operational, detailed scheduling process.
An APS software usually includes functions to manage advanced production plan and resource optimisation processes. APS systems may include the planning of production campaigns, the management of external production by contract companies, cross-plant planning and vendor-managed inventory, among others.
In contrast, a production scheduling software demonstrate its strengths in regard to the operational execution of the production plan. This kind of software facilitates the tracking of material flows and labour utilisation, as well as the provision of real-time feedback on production status.
However, the transition of a production scheduling software and APS is fluid. In terms of an integrated end-to-end approach, all systems should be linked and communicate with each other. An integration into the upstream ERP is also mandatory.
Get in touch!
Would you like to know more about our solutions? Then please write us using the contact form. My colleagues and I look forward to exchanging ideas with you.

Jens Seemuller
Sales Executive