Different industries also mean different requirements for materials and machines, but also for the software. One of the many strengths of our Quality Domain CAQ software lies in the wide range of possible applications in the most diverse sectors. Our satisfied customers include not only companies from the pharmaceutical, consumer goods and electronics industries, but also many well-known automotive manufacturers. Years of experience, innovation and the experience of a large number of satisfied customers have made us what we are today.
Quality Domain - a successful partner to the automotive industry
Initial situation and special features of the automotive industry

In times of global competition, increased safety requirements, the unpredictable development of energy costs, supply bottlenecks and increasing uncertainty due to the growing number of corporate mergers, the remaining market participants are being forced to become more efficient and productive.
Fixed costs must be reduced, which unfortunately often results in a reduction in personnel. This is exacerbated by increasing customer requirements and the individualization of products.
In these difficult times, the use of CAQ software is essential for companies in the automotive industry. It improves the chances of surviving on the global market and helps companies to cope with the ever-increasing competitive pressure.

Strengths of the quality domain

Our Quality Domain software is probably one of the most versatile CAQ software currently available on the German market. It supports you in all areas of your quality assurance and paves the way for a continuous improvement process. By using the software, you can analyze quality-relevant data right at the start of the production process in order to detect key errors, tolerance violations or trends at an early stage.
Thanks to the central storage of all relevant quality data, the traceability of each individual piece of information is possible without great effort – so that, in combination with our complaint management software, the causes can be easily and specifically tracked down and rectified as quickly as possible. With the help of our traceability software, you can find part IDs and batch data more easily and carry out process improvements more efficiently – resulting in increased customer satisfaction.

Dynamic test severity control
An important, because cost-saving, aspect of the software is the dynamic control of inspection accuracy. It is based on the process data that is collected directly during the production process and documented on a daily basis, such as various quality-relevant parameters that welding robots transmit to the software during the welding process. This makes it easy to identify conspicuous welding points so that a targeted inspection can then be carried out that is limited to these conspicuous points. This process drastically reduces the number of inspections and the associated costs.
Handling the software
Another strength of the software is its user-friendliness, which we receive time and again from our customers. This is due to the clear design and self-explanatory functionality. This also includes the quick and easy familiarization of employees with the software. This is done individually according to user groups, so that each employee receives training specifically tailored to their area of responsibility.
In addition to the special training models, the user profiles can also be customized to ensure that the software remains clear for each individual. The inspector is therefore quickly able to enter measurement data, which is then analyzed by the process monitor over a longer period of time in order to detect process changes, such as repeat errors, using long-term analyses.
Continuous adaptation of the software
Joining technologies in the automotive industry have developed rapidly in recent years, with the result that numerous different technologies are currently in use. The integration of new joining technologies into the software is an ongoing process, ensuring that you can always integrate the latest technologies that the market has to offer.
Connection of process and product data
The software enables you to bridge the gap between product data and quality-relevant process data. Not only can all process data collected during production using the various joining techniques be accessed centrally in the software, but also all product information from the various systems, such as ERP or CATIA. With the Report Designer, this data can then be displayed in correlation with each other in meaningful evaluations. The flexible creation of test certificates, weekly and monthly evaluations, measurement reports or capability certificates is also extremely simplified.
The Report Designer offers a variety of standard display formats. If these are not sufficient, you can even create new individual reports with little effort. By integrating graphics, photos and CAD drawings (including MIBA), reports can be adapted to individual requirements. However, the software can not only centrally manage the product and process data mentioned, but of course also all other process data, such as from coordinate measuring machines, gap and transition measuring devices or torque wrenches.

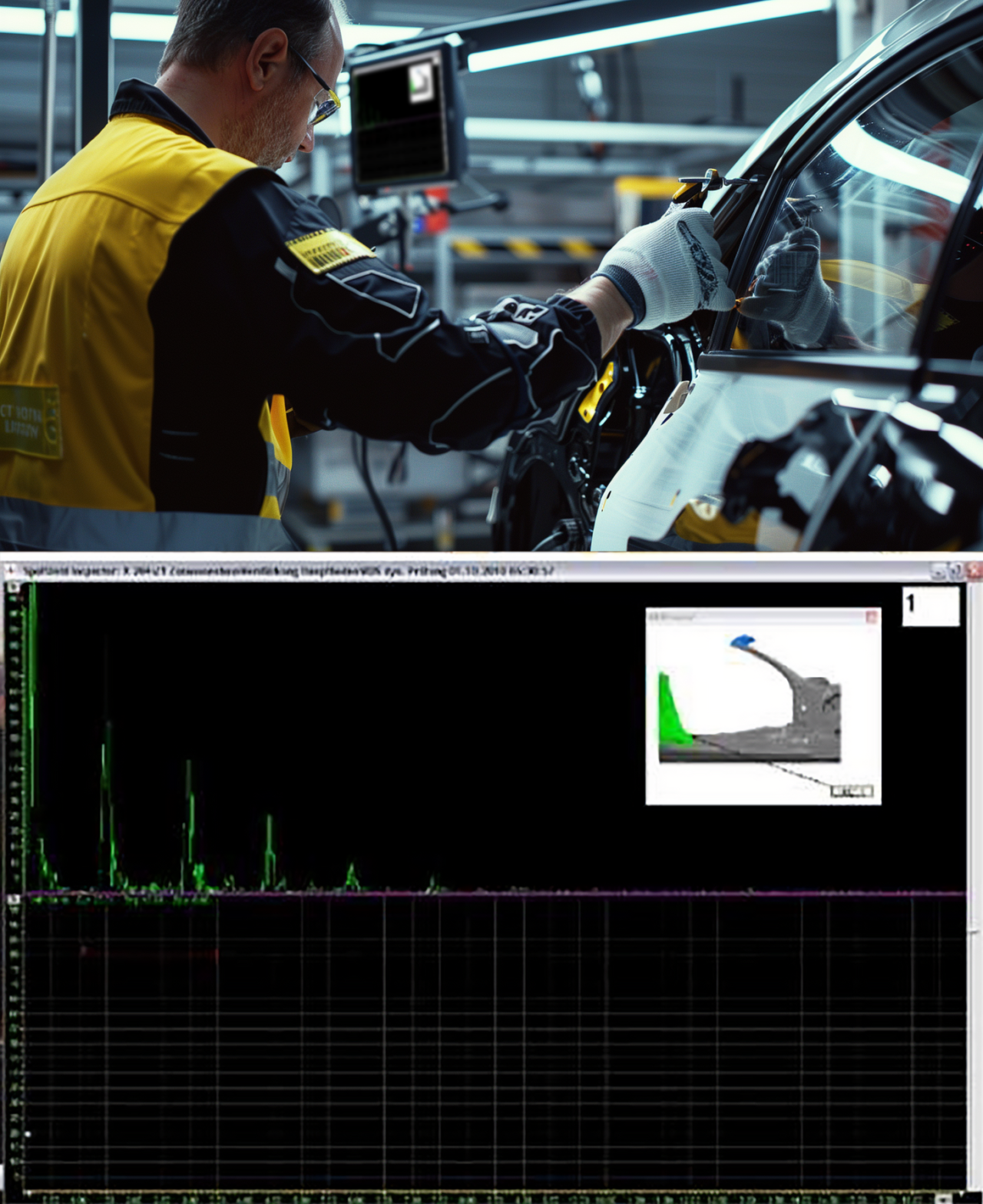
Application example: Testing spot welds with ultrasound
The picture at the top right shows an employee checking spot welds with an ultrasound device. These results are then displayed on a monitor, as shown in the picture below. QDA provides a visual representation with the exact position of the individual measuring points as support for the ultrasonic inspection, which is also displayed as a 3D graphic on the monitor.
Why companies in the automotive industry choose us
Thanks to our global service and short implementation times for software problems or inquiries, we always manage to convince our customers of us and our products and build up a long-term working relationship based on trust. Many customers appreciate the close and good cooperation with our colleagues, who are always able to think their way into the most diverse problems of the respective project. Following a large number of discussions and analyses, optimal solutions are then sought together and individual decisions are made. Thanks to the good contact that continues beyond the project, the software can be continuously and optimally adapted to the constantly changing production processes.
Get in touch!
Would you like to know more about our solutions? Then please write us using the contact form. My colleagues and I look forward to exchanging ideas with you.

Dominik Weggler
Head of Sales Germanedge
Armaments group tightens quality processes with fully integrated eQMS
Our client is a supplier of complex defense systems. As a global player, the OEM works with hundreds of Tier 1 and Tier 2 suppliers. The multinational company has been on a stable growth path for years. As a result, the need to transfer quality management processes, tasks and data to a central planning and control solution increased. In 2012, the company began looking for a suitable QMS solution.
What were the main challenges?

The dominant topic on the agenda of QM managers is traceability. When defense systems are manufactured, a large number of parts and assemblies have to be processed. The requirements for the traceability of these parts extend down to the level of individual measuring points.
In order to cover the entire value chain, data and information must be processed both from the company’s own systems and from key suppliers – from ID and batch numbers to material certificates. Before the launch of QMS, the customer used a framework document that accompanied the components through the entire production process. All process participants were required to document their work in accordance with the company’s QM specifications. However, the increasing complexity of the production processes and the growing number of orders also increased the risk of no longer being able to fully record the required data.


The defense company was therefore looking for an integrated QMS – a data-based software system that enables bidirectional exchange with all relevant systems in the value chain. In particular, this includes the company’s SAP solution, the MES systems used and the measuring instruments. Additional attention is paid to the integration of the calibration system. This ensures that operators only use validated tools for their quality checks. This means that when a measuring device is used, the eQMS automatically checks whether it is correctly calibrated so that its results can be accepted.

Why did the customer choose QMS?
In addition, the entire architecture of the solution, especially the data structure, is transparent enough to be validated by both internal quality managers and external auditors. In this way, it can be proven at any time that the manufacturing processes comply with all regulatory requirements. These range from industry standards such as ISA95, ISO9000 and TFS16949 to customer-specific requirements and IT security and data protection regulations.
How did the implementation process go?

The armaments group operates the software as an on-premise solution in one of its European plants. The basic implementation took place in 2013. The rollout began along the entire value chain. The customer was able to carry out large parts of the implementation itself. This applies in particular to user training. Experience shows that new users are usually up and running within two hours. This is mainly due to the lean GUIs of QMS and not least the fact that the system runs in the local language.


From a process perspective, the implementation began with a proof of concept (PoC). This showed that the customer was already able to map the majority of its processes in the QMS standard. In addition, the PoC made it clear where changes needed to be made. Most of these modifications were necessary to adapt the traceability functions of the software to the requirements of the defense industry. Thanks to the PoC, the customer is still working with a solution where all changes are integrated into the standard.
In addition, virtually no changes were made to the actual core solution after the initial implementation. Adjustments are only made almost exclusively when additional integrations are required. For example, when new production facilities or measuring devices need to be connected to the QMS.

How does the QMS support operational processes?
QMS is used along the entire value chain. The spectrum ranges from incoming goods to delivery control. This ensures that no part leaves production without having been approved in eQMS. The company also uses QMS to approve newly developed parts and new suppliers for practical use.
More than 800 concurrent users work with the system around the clock. The user interfaces have been designed for the various user roles. This means that users only see the data fields and associated QMS information that are relevant to their specific work. The customer has set up these front-end roles themselves.
The armaments group uses the functions of the Quality Domain in these areas in particular:
Together with the Quality Domain, the customer is currently considering how measurement data management can be further optimized. At the center of these considerations is the use of artificial intelligence and natural language processing (NLP). The aim is to build a voice input system that allows workers in the hall to record measured values without having to interrupt their actual work.
How can faults be rectified or even avoided?
Considering the range of applications for its products, it is crucial for the equipment supplier to identify problems, defects and faults in the manufacturing process and even avoid them if possible. This is why the QMS customer has a comprehensive solution for error handling.
The automatic escalation processes of QMS can be triggered even when irregularities are just beginning to emerge: If a measurement trend threatens to deviate from its usual distribution pattern, the system recognizes this at an early stage. This gives the set-up supplier more time to adjust the affected production processes before the produced parts actually fall out of specification.
This enables quality managers and product owners to carry out predictive quality analyses. Although the currently implemented solution does not yet use artificial intelligence for this purpose, the customer is able to manage and control deviations from the specification at a very early stage. This is because whenever an error occurs, the error management system triggers an alarm and automatically notifies the people responsible. They also automatically receive exactly the information they need to rectify the current problem. In this way, the equipment supplier minimizes the number of production errors and, as a result, the amount of its.
Get in touch!
Would you like to know more about our solutions? Then please write us using the contact form. My colleagues and I look forward to exchanging ideas with you.

Dominik Weggler
Head of Sales Germanedge
Customer use case: LIMS at a medical technology company
We can say a lot about ourselves and our software. However, no one can say more about our software than our customers, who use it every day.
Our user reports show how companies achieve success with our solutions. There is no better way to find out what our customers think! The following is a user report on LIMS at a medical technology company
LIMS Use Case
- Overview: International manufacturer of implants for joint replacement surgery uses CAQ software from Quality Domain to save more time in data acquisition in the future.
- Challenge: Compliance with standards and guidelines, as well as recording, managing and analyzing the enormous amount of data.
- Solution: Introduction of QMS goods receipt for immediate visibility and easier handling of data.
- Result: Reduction of errors and time savings in data collection

The challenge: data collection and traceability
The medical industry is under close scrutiny. Many checks have to be carried out and the authorities specify a range of standards and guidelines. Such as DIN EN ISO 13485 or FDA21 CFR Part 11, according to whose specifications the CAQ software must also work.
Furthermore, companies that manufacture medical products are legally obliged to store all their process data for 30 years so that even years later it is still possible to say exactly which patient has had which implant inserted. And how the part was manufactured or with what results. This means that a complete process history must be ensured for every product manufactured over the past 30 years.


Solution: QM incoming goods
The company first started a pilot project and implemented QM software in the incoming goods department. For the first time, the workers were able to carry out their inspections graphically and with text support. The inspection planning was set up identically to the production process. One advantage of computer-aided data recording is the ability to immediately visualize all recorded measurement data.
After successful completion of the pilot project, validation was carried out by the FDA. An external auditor inspected the entire process chain, from development and pilot series to market launch. After successfully completing the validation and realizing the versatility of the software, the customer decided to integrate further QMS modules into their production, such as the SPC software or the Import Manager with the Import Server.
Due to the simple handling of the software, only the training of a quality representative was necessary.


Result: Decades of availability of all data and significant time savings in data acquisition
Thanks to computer support, the customer is now able to display all process-related data for a part at any time. In addition, statistical evaluations can also be carried out retrospectively with just a few mouse clicks.
For certain product families, time savings of 20-30% have even been achieved in data acquisition and a reduction in the susceptibility to errors has been observed.
Thanks to the implementation of QM, the workers are now able to see directly in which area of the specification the currently measured value is located. Trends are therefore easily recognizable, so that an early reaction is possible before tolerance violations can even occur.

Get in touch!
Would you like to know more about our solutions? Then please write us using the contact form. My colleagues and I look forward to exchanging ideas with you.

Dominik Weggler
Head of Sales Germanedge
Customer use case: Wind energy
We can say a lot about ourselves and our software. However, no one can say more about our software than our customers, who use it every day.
Our user reports show how companies achieve success with our solutions. There is no better way to find out what our customers think!
Below is a customer use case from the wind energy sector. With more than 40,000 installed wind turbines, the company is one of the largest wind turbine manufacturers with several production sites worldwide.

Divisions
Each of the four product divisions is dedicated to the manufacture of a specific part of the wind turbine. There are aspects of quality management, such as data acquisition, that affect all divisions. However, there are also individual quality requirements that are only relevant to certain areas.
The nacelle consists of gears and bearings that are built around the main shaft and are driven by the rotor blades. Additional quality requirements here include the collection of torque data on all fasteners, SPC and traceability of components and materials.
The tower is the pillar, the base on which the nacelle is mounted. The recording and analysis of welding data, the documentation of material assemblies and traceability are the top priorities here.

Rotor blades. Each wind turbine requires three rotor blades, which are made of carbon fiber and glass to save weight. Depending on the type of wind turbine, the rotor blades are available in different lengths. The quality focus in the production of the rotor blades is on controlling the weight of each rotor blade and maintaining the exact shape for perfect aerodynamics. The elimination of faults and defects is essential, as the scrapping of a faulty rotor blade is associated with high costs.
Control systems. These are the hardware and software that control the wind turbine. The focus here is on achieving yield targets for the initial samples, supplier management and the tracking of parts and defects.

Global standardization of data collection
Given the size of the company, the complexity of the quality requirements and the global distribution of production sites, reconciling data and reports generated in different plants and by different suppliers is a major challenge. Millions of measurements are taken and analyzed every day.
Our QM software is installed on the central server. This is where all the data is recorded, stored and managed. The software standardizes all quality data. It ensures that measurements of the same characteristic are taken and analyzed in the same way everywhere. Consistent quality management begins with data acquisition. Even here, it must be ensured that all data is valid before it is imported and further processed. This guarantees the comparability of all data from every system and every machine worldwide.

Implementation and quality integration
The first step in introducing a new product or solution to a wind turbine manufacturer is a successful pilot installation. The pilot installation for QMS took place at the company’s headquarters in 2008 and included the QMS SPC software. Data was to be imported from over 500 devices and from various databases. In addition to the company’s own equipment, this also included welding machines, Zeiss coordinate measuring machines and special machines and test equipment for the wind energy sector.
QMS is implemented throughout the entire product life cycle chain, including the planning and approval process for newly developed parts and suppliers across the entire production chain with more than 5,000 employees working in the system around the clock. No part leaves production without being approved in QMS.


The manufacturer utilizes the possibilities of QM for use in:
- Quality planning – APQP4Wind including process flow, inspection plan, FMEA, PPAP and MSA.
- Incoming goods – quality assurance and release of incoming goods, including inspection and documentation, directly integrated with ERP.
- Data collection – Inspection planning to visualize characteristics, provide work instructions and support inspections directly integrated with MES.
- Data analysis & reporting – Visualization and partial acceptance via automated analysis & reporting using statistical methods directly integrated with KPI reporting.
- Complaint Management – Automated integration between data collection and defect resolution has significantly reduced the time required to manage the 8D process

ERP integration
QMS is integrated into the company’s existing SAP system and thus automatically receives further information, such as parts data, supplier master data, information on incoming goods orders or returns. This exchange takes place in both directions and also in exchange with warehouse management.
The MES connection
QMS is also integrated into the company’s existing MES system and also receives its information from here. It automatically receives production orders and recorded values from the MES system and returns relevant quality data to the MES system.
The synergies
QMS ensures that all quality data, from product planning to continuous improvement, is managed in an integrated system. This has created synergies over the years and significantly reduced quality costs. All recorded data, whether from the design phase or production, is error-proof and valid from the outset. It pays off across the entire line when reliable data is recorded. The integrated quality management system can thus get the best out of the data. In addition to the use of quality data analyses, the data can be passed on to suppliers and customers as documentation or product certificates. This forms the basis for quality planning, compliance and inspection, the use of complaint management software, and continuous improvement.
The automated analysis and reporting functions are located along the entire quality value chain and can therefore uncover problems in real time. The graphically supported reports are clear and easy to read and provide the right people with meaningful information to make robust decisions in a timely manner.
Customer focus

The company’s success depends on the successful installation and operation of the wind turbines at its customers. The wind turbine manufacturer takes on an active advisory role when working with its customers in order to develop the special requirements of its customers together with them and to support them with its specialist knowledge throughout the planning phase. The QMS-based quality assurance system is an essential part of the production process.
The software applies demand-oriented production resources for quality control at every process step, defines strict and precise takt times, utilizes SPC with Six Sigma objectives, establishes employee certification processes, and standardizes gauge and equipment management through integrated calibration management software.

Customer-specific analysis

The Quality Domain has developed a process in which work instructions run automatically. The QMS software itself assesses whether the production line is within the quality tolerances and the specified sigma level. QMS analyzes the output of certain processes to determine whether they are being carried out optimally or whether the very time-critical inspection and data acquisition processes need to be adjusted. Another customer-specific module was implemented to obtain automatic test certificates from QMS and to provide test evaluations using the QMS Report Designer.

Gondel Baugruppe
The entire nacelle assembly process is mapped in a single QMS inspection plan, with each step defined as a process. The inspection plan, which includes “order” fields and sample information, requires that each individual step must be completed with an “OK” before the next step can start. It also ensures that each component is traceable. These inspection plans contain up to 450 processes and cover several hundred characteristics.
Today, all information from production flows into the QMS database to ensure that documentation and work instructions are up to date. Once a nacelle has been assembled, including the correct cable attachment, the recorded data is imported using the QMS Import Manager. If QMS determines that the inspection results are OK, it summarizes the results in the corresponding reports. The software then automatically creates and distributes the inspection reports. The process generates millions of records that are stored in QMS. Therefore, QMS is able to log test results, identify defects, track rework and calculate the values that go into KPIs to enable meaningful comparisons.
With QMS at the core of its quality management platform, the company has introduced what all leading manufacturers are trying to achieve – an exemplary integrated quality system that continuously improves quality on multiple levels, reduces costs on a large scale and ensures your product performance worldwide.
Get in touch!
Would you like to know more about our solutions? Then please write us using the contact form. My colleagues and I look forward to exchanging ideas with you.

Dominik Weggler
Head of Sales Germanedge
Seamless global integration of QMS into decentralized ERP infrastructure
Project overview
A global manufacturer of plastic products at over one hundred locations uses our QMS LIMS software to enable cross-location processing of inspection orders. Furthermore, the aim of the LIMS software is to create comparability between the plants through identical evaluations of the same materials or products.
The challenge
The QMS LIMS serves as a system for the laboratory for data acquisition and for quality management and development as a tool for evaluations. The central system is used for all locations of a business unit. The challenge here is to standardize the different processes and philosophies of the locations in one LIMS system. The connection of the decentralized ERP systems also poses a challenge. As part of the project, different IT systems and working methods had to be harmonized in one LIMS.
Solution
The QMS LIMS module is used in direct connection via our module for ERP integration with the decentralized ERP systems. Test planning is mainly carried out in the SAP QM module and the site-specific standards stored there are used. In some cases, several ERP systems communicate with a shared laboratory and its QMS LIMS database.
The QMS solution is used for complete order management, from receipt of the sample, sample preparation, pre-treatment and recording of measured values to the subsequent decision on use. Furthermore, the order planning is followed by an evaluation of the measured values collected in the form of test reports, forms and business intelligence reports.

The respective test devices and systems are addressed directly via the QMS LIMS using uni-respectively bidirectional interfaces and the results of the respective tests are read out using predefined rules. The customer uses various types of interfaces, from file import interfaces to web service interfaces.
It is crucial for the customer that all measuring devices are connected to the QMS LIMS in order to realize fully automatic data acquisition. By connecting the test equipment, the resources of the employees are spared, as they can focus entirely on the day-to-day business. It also optimizes the time required for order processing, as the effort involved in the inspection process is reduced. By connecting test equipment to QMS LIMS, the customer ensures that all tests are controlled by the laboratory and that tests can be compared at any time.


Its use is not limited to the basic function of a LIMS. At many sites, QMS is used to record data for incoming and outgoing goods inspections or for related analyses, such as mixture releases. In addition, QMS is used for the internal laboratory performance of test equipment capability analyses. Thanks to the high degree of configuration options, the data can be reused or reused for complete consistency within the customer’s application.

Result
The fully automated data acquisition leads to a more effective use of capacities in the laboratory. As a result, employees are able to focus on their day-to-day business. The fully integrated and digitalized process provides better transparency across the entire process, as all process steps are mapped via the LIMS. By concentrating on the day-to-day or core business, the throughput times for test orders have been reduced.
The digitalization of laboratory processes enables the customer to have a paperless laboratory. The central storage of all relevant data for a business unit enables global comparability of materials (finished products, raw materials, etc.).
A look into the future
The global roll-out is currently being implemented together with the Quality Domain project team. By reusing generic templates, resources are saved on the customer side and sibling sites can be connected within a few weeks.
Get in touch!
Would you like to know more about our solutions? Then please write us using the contact form. My colleagues and I look forward to exchanging ideas with you.

Dominik Weggler
Head of Sales Germanedge
Customer Use Case: Fiberline
We can say a lot about ourselves and our software. However, no one can say more about our software than our customers, who use it every day.
Our user reports show how companies achieve success with our solutions. There is no better way to find out what our customers think!
Below is a case study from our customer Fiberline Composites.
About Fiberline Composites
Fiberline Composites, headquartered in Denmark, is one of the world’s leading suppliers of composite materials for carbon fiber turbine blades and wind turbine structures for the wind power industry.
Fiberline’s FRP profiles differ from other traditional construction materials such as concrete, steel and wood and offer several unique advantages.
Challenges at Fiberline
The challenges and problems Fiberline faced were primarily the increasing demands of customers for quality documentation and the requirements for Six Sigma capability. The rapid growth of the company also increased the need for an integrated solution to meet the high requirements and ensure the quality of its own products.

In addition, the different systems used for quality in a combination of Excel, Access, legacy systems and ERP system without any integration made for a very labor-intensive search for solutions. This increased the risk of errors.
To overcome these challenges and turn problems into strengths, an external consultant was hired to evaluate the quality system. During this time, Quality Domain prevailed over the competition and has been in use at Fiberline ever since. The decision to use Quality Domain was based on recommendations. Both the internal quality engineers and one of the most important customers from the wind power industry advocated the selection of Quality Domain. Fiberline was looking for a flexible, user-friendly and cost-effective solution, which they found with us.


The way to a solution
But we support Fiberline? The Quality Domain was involved at a very early stage and supported the creation of the RFP in various workshops. We were also involved in the implementation of the system to support Fiberline’s workflows and meet the requirements of customers and external inspectors.
Fiberline uses SPC software, incoming goods inspection, measurement data management software and Quality Domain ERP integration with Axapta, among others. The implementation began with a definition workshop to determine the processes, naming convention and alignment to ensure that the system not only replicated the existing processes but was also designed for global expansion.


After an initial workshop and basic training, the Quality Domain solution supported a pilot implementation of a specific part that mapped the entire process. This was used as a model/template for further implementation.
After the go-live of the initial project, regular brainstorming sessions and solution meetings were held to ensure that Fiberline was implementing the expected optimizations and fine-tuning the process.
After the system had been in place for four years, a re-evaluation was carried out to adapt the solution to the current situation. It was found that the original implementation was very production-oriented, so Fiberline decided to use additional modules for incoming inspection in the future.

Result
The solution meets expectations and additional functions are added as a natural extension for the suppliers. The change from a standalone system to an integrated system saves a lot of time in daily work. In addition, the product certificate creation process has been changed from a manual to a fully automated process. The interface to Axapta is via file transfer with CSV files. Integration with devices takes place via DFQ files.
Paperless production, optimization of the quality process and simplification of daily work are just some of the benefits Fiberline now enjoys.
Conclusion
“Working with the Quqlity Domain solution has been very good, and the experience of the Domain team has helped Fiberline grow. Having just purchased Quality Domain for our next installations as well, we can say that we are very happy with our decision,” says Kim Birk Larsen from Fiberline.
Get in touch!
Would you like to know more about our solutions? Then please write us using the contact form. My colleagues and I look forward to exchanging ideas with you.

Dominik Weggler
Head of Sales Germanedge
Customer use case: use of predictive quality analytics in body-in-white contruction
The topic of predictive quality analytics is playing an increasingly important role in the planning of many companies. When it comes to the concrete implementation of a solution in the customer environment, not only the technology but also a deep understanding of the processes by all those involved is required.
We present a typical use case as an example white predictive quality analysis in body-in-white production in the automobil sector:

Initial situation
In the past, individual inspection orders were generated on the basis of predefined static inspection plans. In SPC data acquisition, either parts are acknowledged as “OK” or “NOK” in relation to the respective inspection order. In order to obtain an overview of the reported defects, all defect information including images is recorded and displayed. In the specific customer case, more than 10 employees are permanently involved in ultrasonic testing.


Solution – AI-based process optimization
In a joint project from 2019, the basis for the implementation of an AI-based process optimization was set up. One of the aims was to support anomaly detection with a defined algorithm and reduce the scope of inspections. The process data is compared with other welding information already in the system for this purpose. This makes it possible to decide in real time whether the welded spot that has been classified as “n.I.O.” is relevant for a manual re-inspection and needs to be re-inspected by an inspector. In this case, the body is directed to a designated inspection station and the inspector can use an overview to check all the anomaly points collected.
The number and effort of manual ultrasonic inspections has been reduced by more than half thanks to the “Predictive Quality Analytics” approach.


Results and benefits
- By integrating “Predictive Quality Analytics” into the existing inspection strategy, the quality of the ultrasonic inspection is significantly increased, as only conspicuous spot welds (anomaly points) are inspected in addition to the existing equipment inspection.
- The integration of “Predictive Quality Analytics” increases the inspection rate to 100% of all welded spots.
- The image database enables a visual representation of the spot welds to be inspected and feedback on the anomaly points.
- As a result, the customer has achieved a significant reduction in manual ultrasonic inspections.

Get in touch!
Would you like to know more about our solutions? Then please write us using the contact form. My colleagues and I look forward to exchanging ideas with you.

Dominik Weggler
Head of Sales Germanedge